The latest wave of depression treatments in 2026 brings new hope for many Americans. Those that have felt “stuck” after trying numerous antidepressants are seeing huge developments in how the Food and Drug Administration (FDA) and major research centers treat depression by using rapid acting, brain targeted types of treatment. Thus, there is now the possibility that individuals that have treatment resistant depression could see a rapid relief of their depressive symptoms. In addition, new forms of treatment will be available to provide relief to those individuals in a much shorter time frame than medication has provided in the past.
The most watched development of a depression treatment is COMP360, a synthetic psilocybin based treatment for individuals suffering from severe depression. COMP360 is reported to be the first of its kind Synthesis for treating depression. The Synthesis of COMP360 recently completed both phase three trials where statistically significant improvements were noted in comparison to placebo.
Individuals treated with a dosage of COMP360 combined with supervised, therapeutic, medical sessions have shown a tremendous reduction of their depressive symptoms with many participants showing reduction in symptoms lasting from weeks to months. If approved for use by the FDA, COMP360 could be one of the first psilocybin derived treatments for depression available for use within the United States; joining existing treatment options such as esketamine.
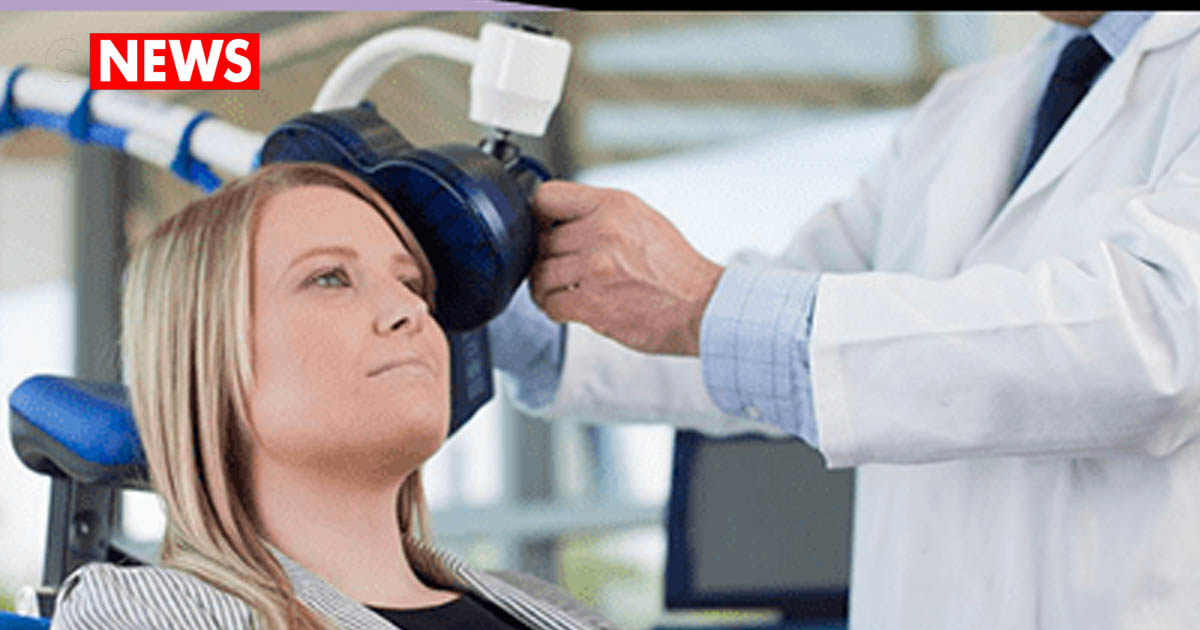
In addition to psychedelic treatments, many areas are emerging for depression treatment via non medicinal forms of treatment, i.e., as device-based treatments. Some of these device-based treatment types include transcranial magnetic stimulation (TMS) and an updated vagus nerve stimulation (VNS) device. Both of these treatments use targeted non-invasive modalities to modulate the brain’s mood control systems. This type of treatment is very attractive for those that do not want the side effects of pharmaceutical medications or want to stop taking medications.
Even with the potential benefits these treatments provide, there are many hurdles to acceptance. For example, many of the newer treatments for depression are very costly and there is no way for some insurance companies to reimburse families for these services under certain circumstances. Additionally, some parents may also have concerns regarding their children receiving “experimental” therapies or how often their children would need to attend a clinic for multiple visits.
Many clinicians who work with depressed children believe that no single method will work for all patients with this disease. In order to help children with depression find long-term recovery, their clinicians typically develop an individualized plan after consulting with the child and/or parent that may include individual therapy, use of medication (when indicated), support groups and changes in lifestyle.
If you are someone who has been through the trials and tribulations of trying different treatments for years and finally found something that helps you feel good again in 2026, you can feel thankful that the use of new technology will help facilitate that experience. Managing the access, understanding, and integration of various treatments towards a holistic plan of compassionate care is the ultimate goal of all who work with the severely mentally ill.
Source: Ubie Health – Newest FDA Approved Treatments for Depression 2026









Leave a Comment